With many competing priorities, it’s not always practical for an early-stage medical device manufacturer or distributor to invest in a CRM system like Salesforce.
Because of this, early-stage device manufacturers & distributors often have person, location, and asset data stored in different silos within the organization. These silos range from email client contacts to documents to spreadsheets.
There comes a point on every medical device company’s growth curve at which the inefficiencies of scattered data become too costly an operational burden.
This is when a medical device company typically begins to evaluate CRM as a shared, central data repository and process automation platform for sales, marketing, customer support, and field service.
Here are ten ways a medical device company can use CRM software to streamline operations and take its business to the next level.
1. Lead Routing and Disposition
Most medical device manufacturers collect website visitor inquiries via a Contact Us form. Often, the input from a form submission is sent to one or more people by email—but not stored in a centralized database. Sales inquiries are manually forwarded to the right salesperson.
When a form submission is a sales inquiry, routing a request to a salesperson via CRM allows salespeople to manage all of their leads in one place. Salespeople can select each lead’s disposition (e.g. disqualified, working, converted) from a list. This is essential feedback for marketing. The fact that leads have a disposition also signals to sales management that leads are getting the proper attention.
2. Medical Device Quoting and Approvals
The format of a medical device company’s sales quotes can range from a simple one-pager to a multi-page document with paragraphs of descriptive text. In addition, the quote may include images.
For some companies, there may be certain quote line items that should only display based on which other line items have been selected. In other words, sales quotes for products are rules-based.
Once quoting requirements are fully defined, CRM can be configured to address those requirements.
If quotes need to be approved before they are sent out, workflows can be created to automate the approval process.
Quotes can be converted to Orders before being sent to the company’s ERP system. This streamlines the entire Quote → Order → Invoice process.
3. Presentation of ERP Data to Salespeople
When a CRM system is integrated with an ERP system such as SAP or Epicor, sales reps have access to customer sales order history within the application they log into daily.
Reports about their customers’ buying activity no longer need to be distributed to salespeople as email attachments.
Customer assets stored in an ERP can also be visible in CRM. For example, a snapshot of a customer’s assets can help guide salespeople’s conversations with their contacts at an organization.
Assets within CRM can be linked to support tickets. By linking assets to tickets, a device’s complete support history can be viewed by any CRM user with appropriate permissions. Problematic field assets can be more easily identified.
4. More Effective & Easily Measurable Digital Marketing
Relevance
Traditional email marketing software is good at getting the word out en masse. But this is often at the expense of targeted, relevant messages to people in specific roles or at a particular stage in the buyer’s journey.
A CRM vendor’s marketing automation system offers a richer customer and patient experience than is available from email marketing systems.
Visibility
Standalone email marketing systems leave salespeople out of the loop regarding what communications their contacts are receiving and how they respond to those messages.
Just as ERP information within CRM benefits salespeople, marketing engagement data gives sales a clearer picture of each customer. Customer and patient engagement with marketing emails can be made visible inside CRM.
Measuring Marketing Campaign Effectiveness
The effectiveness of a marketing campaign is difficult to track when there’s no sharing of activity information between marketing and sales platforms.
When a customer accepts a quote, the associated sales opportunity can be marked as “Closed Won.” The marketing campaign associated with the opportunity can then be credited for the revenue.
With the use of reporting and dashboards, the effectiveness of each marketing campaign can be reported within CRM.
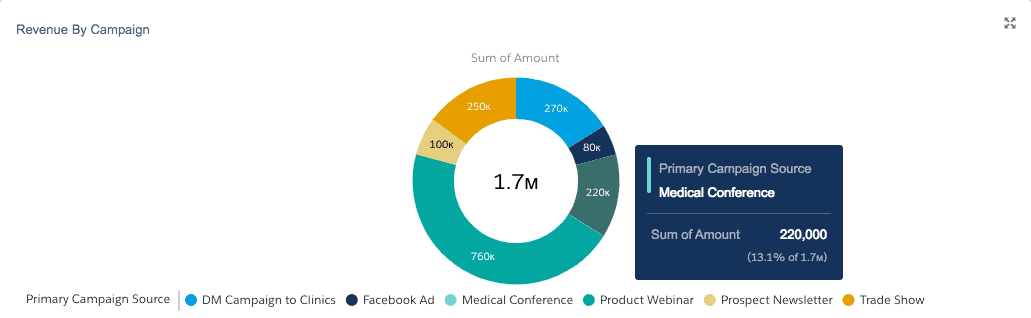
Connecting marketing campaigns to revenue lets marketing leaders determine how to allocate future marketing dollars efficiently.
↓ Article continues ↓
5. Customer Support
For medical device companies, customer service & support issues come in many forms. Support ticket scenarios and resolutions can include:
- Addressing a shipping-related problem
- Training a user who has a knowledge gap in the operation of a product
- Resolving a medical equipment issue via remote diagnostics
- Resolving a device issue via customer hands-on assistance
- Solving an equipment problem with an on-site part replacement
- Entirely replacing a device when no other options are working
No matter what the customer support interaction is, CRM can ensure that issues are resolved promptly. More complex issues can be escalated to engineering or production.
6. Field Service
When an issue with a medical device cannot be solved remotely via human or device interaction, the device may require field service. CRM Field Service functionality facilitates the dispatch of field technicians.
A work order can be created from a support case. A dispatcher can view and manage all appointments in a console. Field service representatives can each manage their day from a mobile phone.
If a field serviceable device is located in a treatment room in a hospital or clinic, field service techs need to know the exact location. This information can be stored in CRM. Each device can have its own physical address.
When devices are wheeled from one treatment room to another, RTLS can be used to pinpoint the current device location for field service purposes.
Within the internet of things realm, medical device data can be fed directly into CRM over a secure connection. This allows for proactive case management and field service. From a revenue perspective, a device IoT connection results in greater patient engagement, which leads to higher retention and cross-selling opportunities.

7. Additional Customer Feedback
Medical device companies constantly seek feedback from healthcare professionals and patients. Traditional ways of collecting feedback are via a questionnaire or a survey. However, surveys may not provide enough feedback. For one, they are not interactive.
A CRM system can make surveys more timely and relevant. For example, a link to a short survey can be sent out immediately after a customer support ticket is closed. The responses to this survey can be attached to their related tickets so the performance of customer service representatives can be measured.
8. Physician, Clinician, and Patient Interaction
A clinical team at a medical device company can benefit from a more free-form and interactive dialog with device users and patients.
The customer-facing functionality can facilitate this interaction. In the medical device industry, CRM-based portals can be configured to serve and interact with anyone outside of the organization who is granted access.
9. Case Tracking and Practitioner Training
CRM can be used to create and track more surgical cases. The CRM can have custom fields so that a salesperson can enter information about procedural case information and other data points specific to a device’s function.
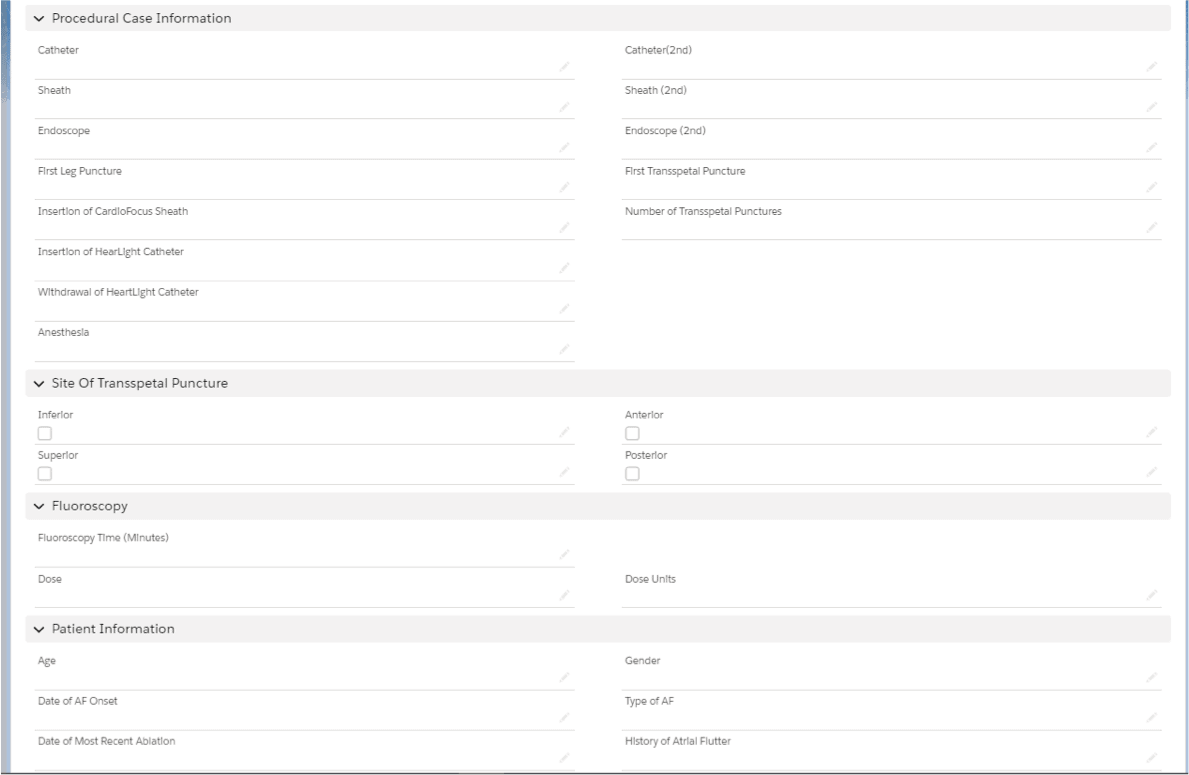
Practitioner training events can be scheduled and marked completed for reference.
10. Reporting and Analytics
There are tools in CRM systems like Salesforce that provide business insights in addition to the above-mentioned marketing effectiveness dashboards.
For example, specific reports give insights into customer satisfaction and identify potential issues before they become significant problems.
AI-driven analytics can look at data from sales opportunities and highlight at-risk deals on a salesperson’s or sales manager’s home screen.
Extended CRM Functionality
Because CRM systems like Salesforce are complete development platforms, implementers can extend them to address needs specific to a medical device company.
By analyzing and prioritizing a device manufacturer’s or a distributor’s essential business requirements, the level of required extended functionality can be determined.
Get more out of your medical device company’s Salesforce implementation with the help of J2’s experts.

